Method Development & Validation
Method Development & Validation Services
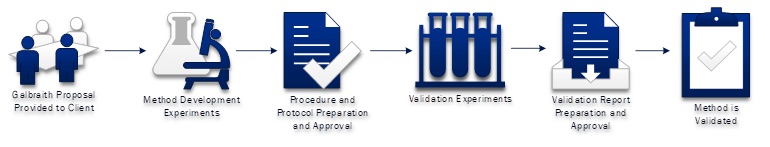
Galbraith Laboratories provides method development and method validation services to clients that have a regulatory need to use a validated procedure or to clients that want the additional assurance the method is working as intended. The purpose of method validation is to demonstrate that the established method is fit for the purpose. These projects begin with a needs-based assessment followed by a formal proposal from Galbraith. The proposal is based on the applicable regulations, product specifications, technical requirements, historical analysis of the sample (if any), and any other client needs.
Method Development
Galbraith begins by performing all validation experiments in a pre-validation, non-regulated mode. The initial methodology used in these experiments can be a general Galbraith procedure, a client procedure, or a draft procedure. This step gives Galbraith the opportunity to optimize the method to meet the necessary requirements, and to establish attainable acceptance criteria for the validation protocol.
Galbraith highly suggests adding some additional stability work to the project requirements so that stability of the drug becomes more well understood. A simple method development can be enhanced to be a full stability indicating method by adding on forced degradation testing. Stability indicating methods adequately demonstrate control of the drug and its degradant products and help produce the most robust methods possible, for improved performance and specifications. See the following link for more information on forced degradation testing.
Forced Degradation Testing
Validation Protocol and Procedure
Once the development work is completed, Galbraith will prepare a custom procedure and protocol. Procedures are written to include a description of all instruments, reagents/standards, and steps required to perform the method for the client’s sample. Protocols are typically designed to meet the ICH and USP requirements, though protocols may be designed to meet other guidance as well. Experiments are usually conducted to measure Accuracy, Repeatability, Intermediate Precision, Specificity, Linearity, Range, Limit of Detection, Limit of Quantitation, and robustness. Once the protocol and procedure have been approved by Galbraith Management, Galbraith Quality Assurance and the client, the validation experiments will be conducted.
Validation Report
Once the validation experiments are completed, the data will be analyzed and a validation report will be prepared. The report will contain all of the validation results, a statistical analysis of the data and overall conclusions regarding the validation of the method. Once completed, the report and all associated data are inspected by Galbraith’s Quality Assurance unit before it is sent to the client for approval. When all approvals have been obtained for the validation report, the method is considered validated and may be used for routine analysis of samples.
Method Development and Method Validation Process
Notice: USP <467> Residual Solvents and USP <232> Elemental Impurities
Due to recent changes by the U.S. Pharmacopeia, all USP <467> Residual Solvents and USP <232> Elemental Impurities analyses must be performed after either verification or validation of the specific material. Depending on your regulatory needs, this may require extensive validation. If you require compliance with either USP <467> or USP <232> for regulatory reasons, please request a consultation with one of our technical experts to ensure that your analysis is handled properly.
